Mounjaro sits in a strange place.
People search Mounjaro because they hear about weight loss, but the medication itself is primarily a type 2 diabetes treatment. The weight-loss story matters, but it only makes sense once you understand what tirzepatide is, how the weekly dose schedule works, and why Zepbound exists as a separate brand.
🔑 Key Takeaways
- Mounjaro is the brand name for tirzepatide, a once-weekly injectable medication used with diet and exercise to improve blood sugar in people with type 2 diabetes.
- It activates both GIP and GLP-1 pathways, which is why it can lower A1C and often reduces appetite and body weight.
- The usual adult schedule starts at 2.5 mg once weekly for 4 weeks, then increases to 5 mg; later increases happen in 2.5 mg steps if needed.
- Mounjaro and Zepbound contain the same active ingredient. Mounjaro is the diabetes brand; Zepbound is the weight-management brand.
- The most common side effects are nausea, diarrhea, decreased appetite, vomiting, constipation, indigestion, and abdominal pain.
This article matches the main search intent behind Mounjaro: what it is, what it is used for, how dosing works, what side effects to expect, how cost and insurance work, and how it compares with Ozempic, Wegovy, and Zepbound.
Mounjaro FDA Boxed Warning
The most serious warning class the FDA issues, and the one every prescriber reviews before writing.
Mounjaro (tirzepatide) carries a boxed warning for thyroid C-cell tumor risk, including medullary thyroid carcinoma (MTC). In rodent studies, tirzepatide produced dose-dependent and treatment-duration-dependent thyroid tumors at clinically relevant exposures. Human causation is not confirmed, but the risk cannot be ruled out.
Mounjaro is contraindicated in people with:
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Known serious hypersensitivity to tirzepatide or any formulation component
Report these symptoms to your doctor immediately: lump or swelling in the neck, persistent hoarseness, persistent trouble swallowing, or shortness of breath.
Mounjaro Lawsuits and Recent FDA Label Updates
Context worth knowing before starting.
Mounjaro and Ozempic are named together in an ongoing multidistrict litigation (MDL) consolidating thousands of cases. Core allegations: inadequate warning about severe gastroparesis (stomach paralysis) and related GI complications. A separate set of complaints addresses NAION (sudden vision loss) linked to GLP-1 drugs. Regulatory timeline:
- September 2023: FDA added ileus (intestinal paralysis) to tirzepatide and semaglutide labels after post-marketing reports
- 2024: FDA reviewed reports of suicidal ideation on GLP-1 drugs, did not establish causation
- 2025: FDA updated Ozempic label with NAION warning; tirzepatide labeling under ongoing class review for the same concern
- 2026: MDL cases continue in federal court, outcomes pending
This does not make Mounjaro definitively unsafe. It means the risk profile at scale is better understood in 2026 than it was at approval in 2022. Severe gastroparesis, ileus, and sudden vision loss remain rare but serious reasons to stop and seek care.
What Is Mounjaro?
Mounjaro is tirzepatide.
Tirzepatide is a dual GIP and GLP-1 receptor agonist made by Eli Lilly. In plain English, it copies two incretin hormone signals that help regulate blood sugar, appetite, stomach emptying, and insulin response after meals.
Mounjaro is used along with diet and exercise to improve blood sugar in adults and children 10 years of age and older with type 2 diabetes. It is a prescription medication, not an over-the-counter supplement or peptide product.
The same active ingredient is also sold as Zepbound. That separation creates a lot of confusion. Mounjaro is the diabetes brand. Zepbound is the brand specifically positioned for chronic weight management and obstructive sleep apnea in eligible patients.
Simple version
If your question is "what is Mounjaro?" the shortest answer is this: Mounjaro is a weekly tirzepatide injection for type 2 diabetes that also tends to reduce appetite and body weight.
Mounjaro Uses: What It Is Prescribed For
The main use is blood sugar control.
Mounjaro is prescribed for type 2 diabetes when a healthcare provider decides tirzepatide fits the patient's blood sugar goals, medication history, risk factors, and insurance situation. Lilly reports that in studies, 75% to 90% of adults taking 5 mg, 10 mg, or 15 mg reached an A1C under 7%, depending on dose and study design.
Weight loss is part of the conversation because it often happens during treatment. Lilly states that Mounjaro is not a weight loss drug, but adult study participants lost weight while taking it, with reported average losses ranging from 12 lb at 5 mg to 25 lb at 15 mg in certain diabetes studies.
For people seeking tirzepatide specifically for weight management, the more direct brand to discuss with a prescriber is Zepbound. For broader context on this drug class, see our plain-English GLP-1 explainer.
How Mounjaro Works
Two hormone signals are involved.
Mounjaro activates GLP-1 and GIP receptors. GLP-1 activity helps increase glucose-dependent insulin release, reduce glucagon when blood sugar is high, slow stomach emptying, and increase fullness. GIP activity adds a second incretin pathway that appears to strengthen the metabolic effect compared with GLP-1 alone.
That dual mechanism is why Mounjaro is often compared with Ozempic. Ozempic is semaglutide, which targets GLP-1. Mounjaro is tirzepatide, which targets GLP-1 plus GIP. For a direct head-to-head breakdown, use our Mounjaro vs Ozempic comparison.
In the SURPASS-2 trial, Mounjaro 10 mg and 15 mg lowered HbA1c more than semaglutide 1 mg over 40 weeks. Body weight also fell more with higher-dose Mounjaro in that trial, with the 15 mg group losing about 11.2 kg compared with 5.7 kg for semaglutide 1 mg.
Mounjaro Dosage and Weekly Schedule
The dose starts low.
The typical adult starting dose is 2.5 mg injected under the skin once weekly. After 4 weeks, the dose usually increases to 5 mg once weekly. If more blood sugar control is needed, the dose can increase in 2.5 mg steps after at least 4 weeks on the current dose.
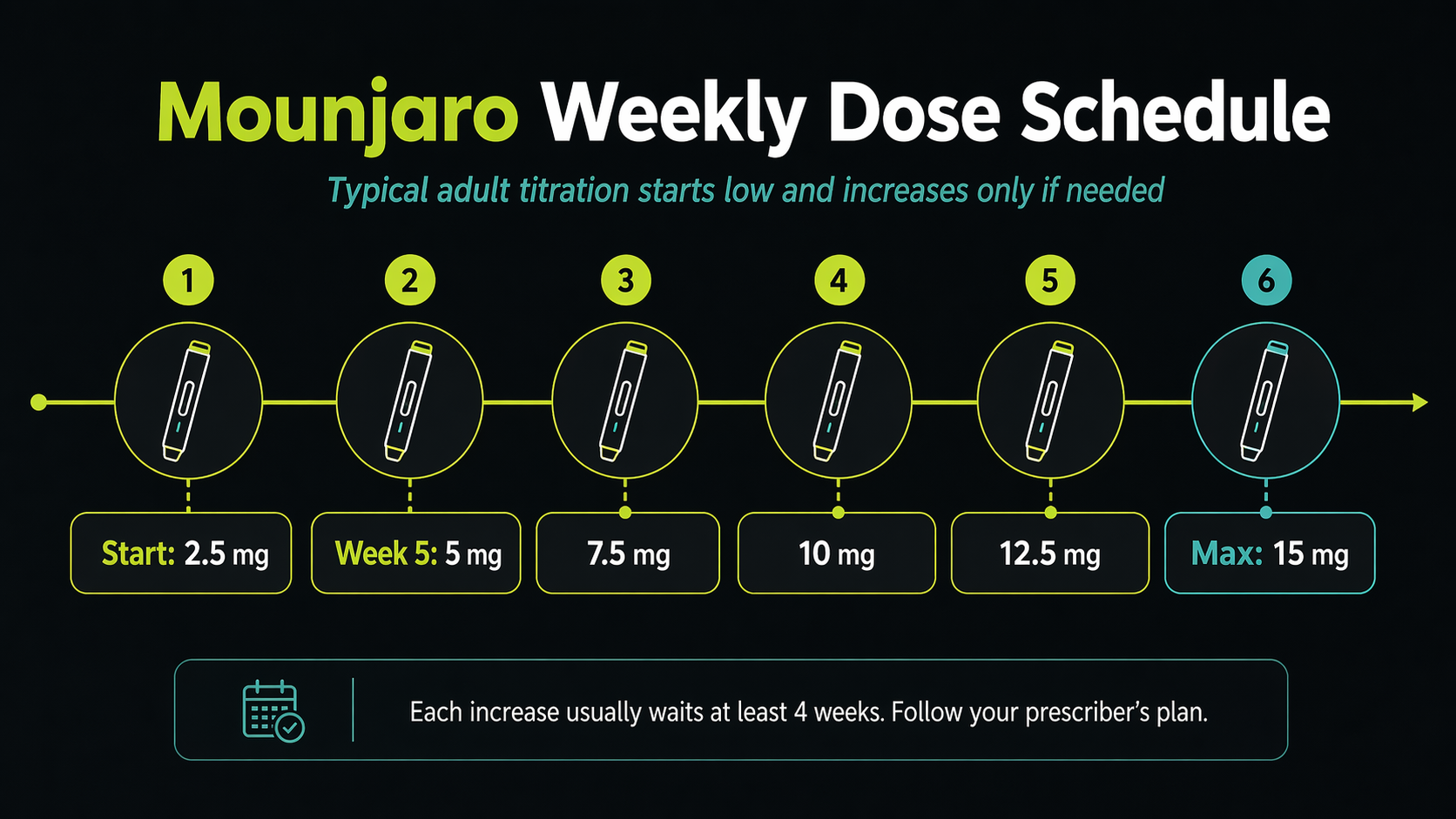
| Step | Typical Adult Dose | How Long | Purpose |
|---|---|---|---|
| Start | 2.5 mg once weekly | 4 weeks | Starter dose to help the body adjust |
| First treatment dose | 5 mg once weekly | At least 4 weeks | Initial maintenance dose |
| Optional increase | 7.5 mg once weekly | At least 4 weeks | If more control is needed |
| Optional increase | 10 mg once weekly | At least 4 weeks | Higher maintenance option |
| Optional increase | 12.5 mg once weekly | At least 4 weeks | If tolerated and needed |
| Adult maximum | 15 mg once weekly | Prescriber-directed | Highest labeled adult dose |
For children 10 years and older with type 2 diabetes, the labeled maximum is 10 mg once weekly. Pediatric dosing should be managed closely by a qualified clinician.
Mounjaro can be injected in the abdomen, thigh, or back of the upper arm when another person gives the injection. Rotate injection sites. It can be taken at any time of day, with or without food.
What If You Miss a Mounjaro Dose?
The four-day rule matters.
If a dose is missed, Lilly and the prescribing information say to take it as soon as possible within 4 days, or 96 hours, after the missed dose. If more than 4 days have passed, skip that dose and take the next one on the regular schedule.
Do not take two doses within 3 days of each other. If missed doses keep happening, the problem is usually schedule design, not willpower. Pick a weekly day that lines up with a stable routine.
Mounjaro Side Effects
The stomach usually complains first.
The most common Mounjaro side effects are gastrointestinal: nausea, diarrhea, decreased appetite, vomiting, constipation, indigestion, and abdominal pain. In pooled adult placebo-controlled trials, nausea occurred in 12% to 18% of Mounjaro users depending on dose, compared with 4% on placebo. Diarrhea occurred in 12% to 17%, compared with 9% on placebo.
| Side Effect | Placebo | Mounjaro 5 mg | Mounjaro 10 mg | Mounjaro 15 mg |
|---|---|---|---|---|
| Nausea | 4% | 12% | 15% | 18% |
| Diarrhea | 9% | 12% | 13% | 17% |
| Decreased appetite | 1% | 5% | 10% | 11% |
| Vomiting | 2% | 5% | 5% | 9% |
| Constipation | 1% | 6% | 6% | 7% |
| Abdominal pain | 4% | 6% | 5% | 5% |
These side effects are one reason Mounjaro starts at 2.5 mg. The goal is to give your digestive system time to adapt before the dose climbs.
Call a healthcare provider promptly for severe or persistent abdominal pain, repeated vomiting, signs of dehydration, symptoms of gallbladder trouble, severe allergic symptoms, or low blood sugar symptoms if Mounjaro is used with insulin or a sulfonylurea.
NAION (Sudden Vision Loss), 2025 Class Review
Research in 2024-2025 linked GLP-1 drugs to a higher risk of non-arteritic anterior ischemic optic neuropathy (NAION), a sudden and usually painless vision loss in one eye caused by blocked blood flow to the optic nerve. The FDA added NAION to Ozempic's label in 2025, and tirzepatide is under ongoing class review. Any sudden vision change, especially in one eye, needs same-day ophthalmology evaluation.
Ileus (Intestinal Paralysis)
Added to Mounjaro's label in September 2023 after post-marketing reports. Symptoms: no bowel movements for several days, severe abdominal distension, persistent vomiting. Ileus is a medical emergency, stop the drug and go to the ER.
Acute Kidney Injury from Dehydration
Persistent vomiting or diarrhea can cause dehydration severe enough to trigger acute kidney injury, especially in older adults and those with pre-existing kidney disease. Stay hydrated aggressively during dose escalation; call your doctor if you cannot keep fluids down for 24+ hours.
Warnings and Who Should Avoid Mounjaro
Some people should not use it.
Mounjaro is contraindicated in people with a personal or family history of medullary thyroid carcinoma, and in people with Multiple Endocrine Neoplasia syndrome type 2. It should also be avoided by anyone who has had a serious allergic reaction to tirzepatide or any ingredient in Mounjaro.
Important warning areas include pancreatitis, low blood sugar when combined with insulin or insulin secretagogues, serious allergic reactions, dehydration that can lead to kidney problems, severe stomach problems, gallbladder problems, and vision changes in people with diabetes.
Mounjaro can also reduce the effectiveness of birth control pills because it slows stomach emptying. The prescribing information advises switching to a non-oral method or adding a barrier method for 4 weeks after starting Mounjaro and for 4 weeks after each dose increase.
Mounjaro for Weight Loss vs Zepbound
This is the common confusion.
Mounjaro and Zepbound are both tirzepatide. The dose strengths overlap. The difference is the labeled use and how insurance tends to process the prescription.
| Brand | Active Ingredient | Main Use | Why It Matters |
|---|---|---|---|
| Mounjaro | Tirzepatide | Type 2 diabetes | Usually covered through diabetes benefits when criteria are met |
| Zepbound | Tirzepatide | Weight management and related indications | Usually routed through obesity-medication coverage rules |
If someone does not have type 2 diabetes and wants tirzepatide for weight management, Zepbound is usually the cleaner conversation with a provider. Mounjaro may be prescribed off-label in some cases, but insurance often pushes back when there is no type 2 diabetes diagnosis.
For the broader weight-loss injection landscape, including semaglutide, tirzepatide, and retatrutide, see our weight loss injections comparison.
Mounjaro Cost, Insurance, and Savings
The sticker price is high.
Mounjaro commonly costs around $1,000 or more per month before insurance or savings programs, depending on pharmacy and plan details. The real price a patient pays can be very different because coverage, deductibles, prior authorization, and savings-card eligibility all matter.
Lilly's savings information says eligible commercially insured patients with Mounjaro coverage may pay as little as $25 for a 1-month, 2-month, or 3-month fill. For eligible commercially insured patients without Mounjaro coverage, Lilly lists a lower cash pathway that may be as low as $499 for a 1-month fill, subject to program rules and expiration dates.
Government insurance programs have different rules. Medicare, Medicaid, VA, TRICARE, and other government beneficiaries are generally excluded from manufacturer savings-card programs. If cost is the blocker, start with the prescriber, pharmacy, insurer, and Lilly's current savings terms before assuming the retail price is final.
For more ways people compare GLP-1 access, read our GLP-1 without insurance cost options.
How to Get Mounjaro
You need a prescription.
The usual path is a visit with a primary care doctor, endocrinologist, obesity medicine clinician, or qualified telehealth provider. The provider may review A1C, fasting glucose, weight history, current medications, kidney function, digestive history, pregnancy plans, and insurance requirements.
Once prescribed, Mounjaro is filled through a pharmacy. Availability can vary by dose, location, and insurer. If one dose is unavailable, do not improvise by doubling a lower dose or changing the schedule on your own. Ask the prescriber and pharmacy what dose substitutions are appropriate.
Before the first fill
Ask your prescriber three things: what dose you are starting on, when the dose might increase, and what side effects should trigger a same-day call.
Mounjaro vs Ozempic, Wegovy, and Retatrutide
The names blur together fast.
Mounjaro is tirzepatide. Ozempic and Wegovy are semaglutide. Retatrutide is a newer triple-agonist still being studied for broader use. The practical difference is not just "which one is strongest." It is the active ingredient, labeled use, insurance pathway, side effect tolerance, and what outcome the prescriber is targeting.
| Drug / Brand | Active Ingredient | Main Receptor Activity | Common Context |
|---|---|---|---|
| Mounjaro | Tirzepatide | GIP + GLP-1 | Type 2 diabetes |
| Zepbound | Tirzepatide | GIP + GLP-1 | Weight management |
| Ozempic | Semaglutide | GLP-1 | Type 2 diabetes |
| Wegovy | Semaglutide | GLP-1 | Weight management |
| Retatrutide | Retatrutide | GLP-1 + GIP + glucagon | Emerging next-generation option |
If you want the next layer of detail, the most useful reads are our Wegovy vs Mounjaro and Retatrutide vs Mounjaro comparisons.
Storage and Travel Rules
Temperature control is simple.
Store Mounjaro in the refrigerator at 36°F to 46°F, or 2°C to 8°C. Keep it in the original carton to protect it from light. If needed, a pen or vial can be kept at room temperature up to 86°F, or 30°C, for up to 21 days.
Do not freeze Mounjaro. If a pen or vial has been frozen, it should not be used. When traveling, the practical goal is to avoid freezing, overheating, direct sun, and long periods outside the allowed temperature range.
What to Ask Before Starting Mounjaro
Better questions prevent surprises.
- Why Mounjaro instead of another option? Ask whether the priority is A1C, weight, medication simplicity, or tolerability.
- What dose plan are we following? Know whether the plan is to stay at 5 mg or keep increasing.
- What side effects should I expect? Nausea, diarrhea, constipation, and low appetite are common early issues.
- What should I do if I cannot eat enough? Appetite reduction can become too strong for some people.
- How will insurance handle it? Ask about prior authorization before the prescription reaches the pharmacy.
- What happens if my dose is out of stock? Have a backup plan before refill week.
Mounjaro in 2026: Supply, Price, and What Changed
The Mounjaro supply story is finally boring, which is a good thing. The FDA declared the tirzepatide shortage fully resolved in October 2024, and as of May 2026, every dose of the Mounjaro KwikPen is shipping at retail pharmacies in the US with no allocation limits. That stability changed three things worth knowing about.
First, compounded tirzepatide is no longer a legal workaround. Pharmacies can compound tirzepatide only for an individual patient who needs a clinically meaningful change to the FDA-approved product (a different dose, an allergy to an inactive ingredient, etc.). Mass-produced compounded tirzepatide from telehealth platforms has effectively disappeared. See our tirzepatide compounding pharmacy guide for what is actually still available.
Second, Eli Lilly launched LillyDirect Self-Pay vials for both Mounjaro and Zepbound. For Mounjaro specifically, the single-dose vial program is targeted at patients without commercial insurance and runs $349 to $499 per month depending on the dose. Pricing is sometimes lower for the lower doses. The vials require manual draw with a syringe, so they are cheaper but slightly less convenient than the KwikPen.
Third, insurance coverage for type 2 diabetes is broad and stable in 2026. Most commercial plans cover Mounjaro for diabetes with a copay between $25 and $100, and the Lilly savings card can drop eligible commercial copays to as low as $25. Medicare Part D covers Mounjaro for diabetes but not for weight loss, which still requires Zepbound and the new Medicare GLP-1 Bridge program if applicable.
SURMOUNT-5 and What It Means for Mounjaro Users
SURMOUNT-5 is the head-to-head trial that compared tirzepatide (15 mg) to semaglutide (2.4 mg) in adults with obesity but without diabetes. After 72 weeks, the tirzepatide group lost an average of 20.2% of body weight versus 13.7% for semaglutide. Although the trial used the Zepbound label dose, the molecule is identical to Mounjaro, so the data informs what to realistically expect on the diabetes brand at the same dose.
Two things matter for Mounjaro patients reading SURMOUNT-5 data. People with type 2 diabetes typically lose less weight than people without diabetes on the same dose, often by about a third. So a Mounjaro user at 15 mg should plan for closer to 13% to 16% of body weight lost over a year, not the 20%-plus seen in nondiabetic SURMOUNT participants. And blood sugar improvement is the headline benefit for Mounjaro: in SURPASS trials, A1C dropped an average of 1.8% to 2.4% across doses, which is one of the largest reductions seen for any non-insulin diabetes drug.
For a full breakdown of how Mounjaro stacks up against semaglutide, see tirzepatide vs semaglutide. If you are weighing the weight-loss-only option, our Zepbound vs Mounjaro guide walks through which brand fits each scenario.
Is "Monjaro" the same as Mounjaro?
Yes. "Monjaro," "Manjaro," and "Mounjauro" are all common misspellings of Mounjaro, the Eli Lilly brand name for tirzepatide. There is one real medicine, spelled M-o-u-n-j-a-r-o, and it is the same drug no matter how a search engine autocompletes it.
The spelling matters for one reason: safety. If a website advertises "Monjaro" with no prescription required, or at a price that looks too good to be true, treat it as a red flag for a counterfeit. The only legitimate ways to get tirzepatide are a licensed pharmacy with a valid prescription, Lilly's own LillyDirect service, or a licensed telehealth provider. To lower your cost the right way, see our guide to the Mounjaro coupon and savings card. If your dose is hard to find, check current tirzepatide supply and availability, and if you are fighting your insurer, see how Zepbound and Mounjaro coverage works.
Frequently Asked Questions
References
- Eli Lilly. Mounjaro official patient information. https://mounjaro.lilly.com/
- U.S. Food and Drug Administration. Mounjaro prescribing information, revised 2025. FDA label PDF
- WebMD. Mounjaro (tirzepatide): uses, side effects, interactions, warnings, and dosing. WebMD
- Mayo Clinic. Tirzepatide subcutaneous route: side effects and dosage. Mayo Clinic
- Cleveland Clinic. Tirzepatide injection. Cleveland Clinic
- U.S. News Health. Mounjaro for weight loss: results, side effects, and how it works. U.S. News Health


