In the SURMOUNT-1 trial, up to 45% of people on the 15mg tirzepatide dose reported nausea, making it the most common reason people reduce their dose or stop entirely.
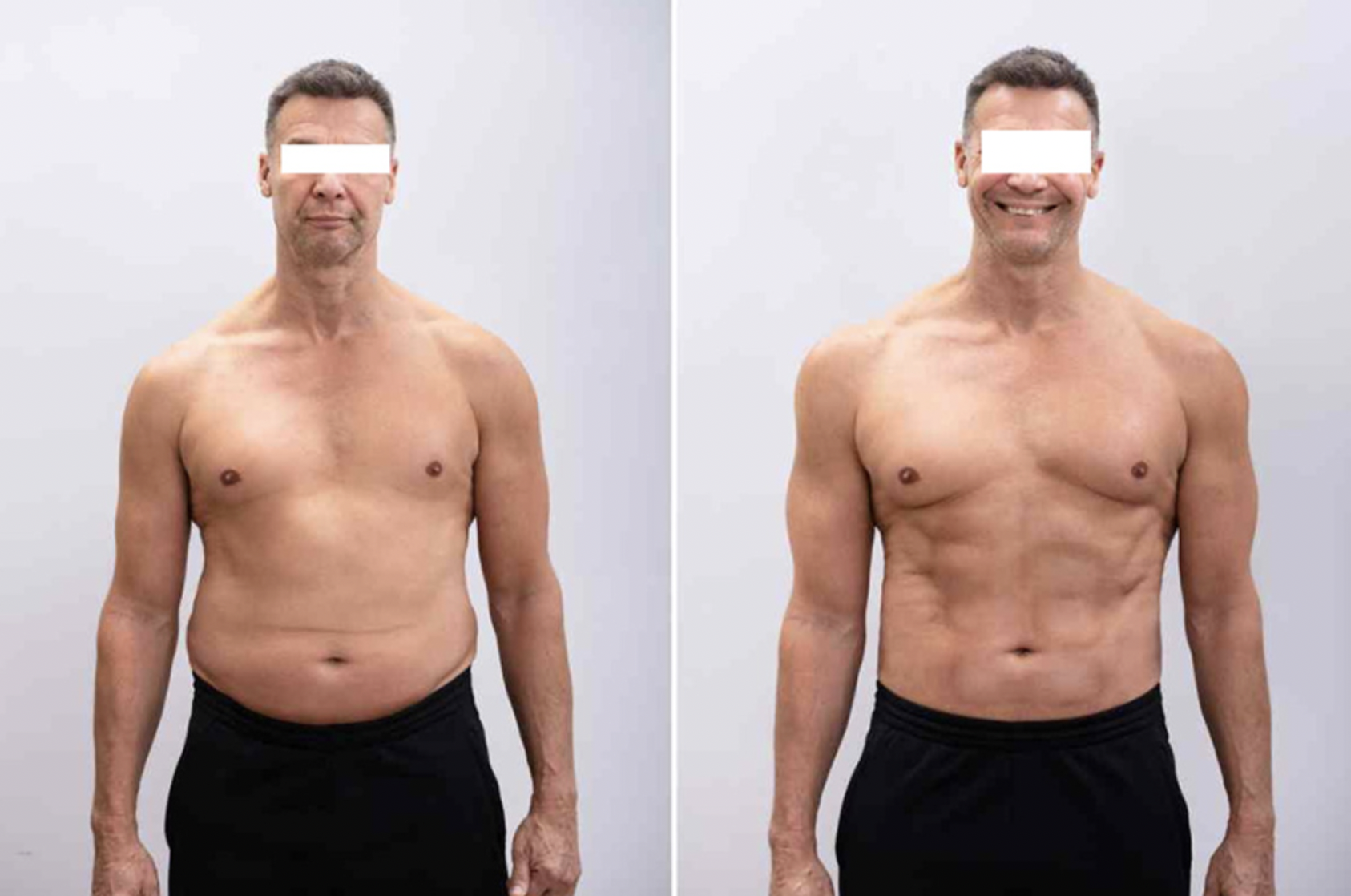
Real GLP-1 before and after results
Four real before-and-after photos from users online who shared their GLP-1 results. Identifiers blurred for privacy. Click any photo to expand.
Photos sourced from users online who publicly shared their GLP-1 results. All four used compounded semaglutide or tirzepatide, the same medications available through MEDVi and Yucca Health telehealth. Individual results vary; trial average is 15-20% body weight loss at 60+ weeks.
🔑 At a Glance
- Most common: Nausea (25-45%), diarrhea (20-30%), constipation (16-29%), vomiting (9-25%)
- Dose-dependent: GI side effects are worse at higher doses and during escalation phases
- Usually temporary: Most GI effects improve within 4-8 weeks as your body adapts
- Black box warning: Thyroid C-cell tumors seen in early toxicology testing, avoid if personal/family history of MTC
- Serious but rare: Pancreatitis, gallbladder disease, and gastroparesis are real risks (<1%)
- Vs semaglutide: Tirzepatide has similar or slightly higher GI rates but substantially better weight loss outcomes
- Stop signals: Severe abdominal pain, persistent vomiting, vision changes, allergic reactions
Tirzepatide's side effect profile is manageable for most people, but only if you know what's coming and how to deal with it. The GI stuff is real, it's annoying, and it does peak during dose increases. This guide covers every documented effect with actual clinical percentages, a dose-by-dose breakdown, and a comparison to semaglutide and retatrutide so you know where tirzepatide stands in the GLP-1 field.
Tirzepatide FDA Boxed Warning
The most serious warning class the FDA issues.
Tirzepatide (sold as Mounjaro for type 2 diabetes and Zepbound for weight management) carries a boxed warning for thyroid C-cell tumor risk, including medullary thyroid carcinoma (MTC). in early toxicology testing, tirzepatide caused dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures. Human causation is unproven but cannot be ruled out. The drug is contraindicated in people with:
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Known serious hypersensitivity to tirzepatide or any formulation component
Symptoms to watch for and report immediately: lump or swelling in the neck, hoarseness that does not resolve, persistent trouble swallowing, or shortness of breath.
How Common Are Tirzepatide Side Effects? Meta-Analysis Frequency Data
The most reliable view of tirzepatide side effects comes from the 2023 systematic review and meta-analysis (Alzahrani et al., Cureus, PMC10614464), which pooled 4,586 patients across six randomized controlled trials. These are the pooled tirzepatide-arm incidence rates vs comparator (placebo, semaglutide, dulaglutide, or insulin):
| Side effect | Tirzepatide incidence | Comparator incidence | Risk ratio (95% CI) | Significance |
|---|---|---|---|---|
| Nausea | 20.43% | 10.47% | 2.90 (1.89–4.44) | P≤0.00001 |
| Diarrhea | 16.24% | 8.63% | 2.07 (1.60–2.68) | P≤0.00001 |
| Decreased appetite | 9.64% | 2.88% | 5.04 (3.01–8.45) | P≤0.00001 |
| Vomiting | 9.05% | 4.86% | 2.69 (1.67–4.36) | P≤0.0001 |
| Dyspepsia | 7.13% | 3.31% | 2.52 (1.58–4.01) | P≤0.0001 |
| Constipation | 2.54% | 0.86% | 3.08 (1.83–5.20) | P≤0.0001 |
What this tells you: tirzepatide side effects in the gastrointestinal cluster are 2 to 5 times more frequent than placebo or comparator drugs, but absolute rates are still in the 2 to 20 percent range — not the majority of users. The decreased-appetite ratio (5x vs comparator) is the most extreme, which is also the mechanism that drives the weight loss in the first place. The trial-specific numbers in the next section break this down further by dose (5mg / 10mg / 15mg) since incidence scales with dose level.
Common Tirzepatide Side Effects (With SURPASS/SURMOUNT Trial Data)
Most tirzepatide side effects hit the gastrointestinal system. This isn't surprising, GIP and GLP-1 receptors both exist in the gut wall, and activating them simultaneously slows gastric emptying and changes bowel motility. Below are the rates pulled directly from the SURMOUNT-1 and SURPASS-2 trials.
| Side Effect | Any Tirzepatide (%) | Placebo (%) | Timing |
|---|---|---|---|
| Nausea | 25-45% | 7-9% | Peaks weeks 1-8 |
| Diarrhea | 20-30% | 10-12% | Ongoing, dose-dependent |
| Constipation | 16-29% | 6-10% | Often alternates with diarrhea |
| Vomiting | 9-25% | 2-4% | Dose escalation phases |
| Decreased appetite | 20-39% | 5-8% | Persistent throughout |
| Abdominal pain | 9-12% | 5-7% | Variable |
| Dyspepsia/GERD | 5-16% | 3-6% | Worse lying down |
| Burping/flatulence | 7-12% | 2-4% | Dose-dependent |
| Injection site reaction | 3-7% | 2-3% | First several injections |
| Fatigue | 8-14% | 5-8% | Early weeks |
| Increased heart rate | +2-4 bpm | minimal | Persistent |
| Hair loss | 4-6% | ~1% | Months 2-5 (telogen effluvium) |
GI Side Effects: Nausea, Vomiting, Diarrhea, What's Actually Happening
The gut is where tirzepatide does most of its work, and most of its damage. Gastric emptying slows significantly, which is partly why you eat less (food sits in your stomach longer, so you stay full). But it's also why nausea is so common. You're not sick; your stomach is just processing everything at a fraction of its normal speed.
Nausea
The most common complaint. It typically peaks in the first 1-4 weeks at a new dose level and fades substantially by week 8. Eating too fast, eating large meals, or lying down after eating makes it dramatically worse. Some people describe it as a low-grade constant background feeling rather than acute nausea.
Vomiting
Less common than nausea but more disruptive. The 15mg dose produces vomiting in roughly 1 in 4 people during escalation. If you're vomiting more than twice per day for more than 2 days at a dose level, that's a signal to hold the escalation and talk to your doctor about staying at the current dose longer.
Diarrhea and Constipation
Odd combination, right? Both happen. Some people get alternating diarrhea and constipation throughout treatment. The mechanism is disrupted gut motility, tirzepatide doesn't uniformly speed up or slow down the bowel, it changes the rhythms unpredictably. Staying hydrated helps. Fiber supplementation helps more. Some people find that probiotics reduce both symptoms.
Injection Site Reactions
These show up in 3-7% of people and are almost always mild. Classic presentation: a small red, raised area at the injection site that's mildly tender for 1-3 days after injection. Rarely itchy. Almost never needs treatment.
Harder, more pronounced lumps can appear if you inject into the same spot repeatedly, this is lipohypertrophy, and it can impair absorption. Rotate injection sites consistently. The approved sites are the abdomen, thigh, or upper arm, rotate through all three across different weeks.
Tirzepatide Side Effects by Dose Level (5mg / 10mg / 15mg)
The dose-dependence is real and consistent across trials. Starting low isn't just about comfort, it gives your body time to build tolerance to the GI effects before moving to higher doses. Skipping straight to 10mg or 15mg dramatically increases the chance of early discontinuation.
| Side Effect | 5mg | 10mg | 15mg |
|---|---|---|---|
| Nausea | ~25% | ~33% | ~45% |
| Diarrhea | ~20% | ~22% | ~30% |
| Constipation | ~16% | ~27% | ~29% |
| Vomiting | ~9% | ~16% | ~25% |
| Decreased appetite | ~20% | ~28% | ~39% |
| Abdominal pain | ~9% | ~10% | ~12% |
| GERD/dyspepsia | ~5% | ~10% | ~16% |
| Injection site | ~3% | ~5% | ~7% |
| Discontinuation due to GI AEs | ~3% | ~5% | ~8% |
One thing that doesn't get enough attention: the 4-week dose escalation schedule is a minimum, not a target. If you're still experiencing significant GI symptoms at week 4, there's no clinical reason you can't stay at the current dose for another 4 weeks before stepping up. The SURMOUNT trials showed good weight loss at all dose levels, see the full tirzepatide dosage chart for protocol details.
Serious Tirzepatide Side Effects
Rare doesn't mean impossible. These are the side effects that warrant a trip to the ER or an immediate call to your doctor, not wait-and-see management.
Thyroid C-Cell Tumors (Black Box Warning)
Tirzepatide carries a black box warning for medullary thyroid carcinoma (MTC) based on early toxicology testing showing C-cell tumors with chronic GLP-1 receptor agonist exposure. Importantly: the human relevance of this finding is unknown. No human cases of tirzepatide-induced MTC have been established in clinical trials.
Pancreatitis
Acute pancreatitis is a class effect of GLP-1/GIP receptor agonists. Incidence in trials was very low (<0.5%), but pancreatitis is serious. Symptoms: persistent severe abdominal pain that may radiate to the back, often with nausea and vomiting. If this sounds like what you're experiencing, stop taking tirzepatide and seek immediate medical care. Don't wait to see if it gets better.
Gallbladder Disease
Rapid weight loss itself increases gallstone risk, and tirzepatide causes significant weight loss. The SURMOUNT trials reported gallbladder-related adverse events in about 1-2% of participants, including cholelithiasis (gallstones) and cholecystitis (gallbladder inflammation). Symptoms: right upper quadrant pain, pain after fatty meals, fever with abdominal pain. Gallbladder issues may require surgery.
Hypoglycemia
Tirzepatide alone rarely causes meaningful hypoglycemia in people without diabetes. The risk is real if you're combining it with insulin or sulfonylureas (like glipizide). In the SURPASS-5 trial with insulin-treated patients, symptomatic hypoglycemia occurred in ~19% of tirzepatide users vs ~10% on placebo. The fix is usually dose reduction of the insulin, not stopping tirzepatide.
Gastroparesis and Bowel Obstruction
The FDA has received reports of severe gastroparesis (stomach paralysis) and bowel obstruction in GLP-1 agonist users. These are rare and the causal relationship isn't fully established, but they're worth knowing about. Red flags: food staying undigested for days, inability to tolerate any oral intake, severe persistent nausea even when not eating. Requires hospital evaluation.
Severe Allergic Reactions
Angioedema (swelling of face, lips, throat) and anaphylaxis are rare but documented. Stop tirzepatide immediately and call emergency services if you experience throat tightening, difficulty breathing, or rapid-onset facial swelling after injection.
Acute Kidney Injury from Dehydration
Persistent vomiting or diarrhea can cause dehydration severe enough to trigger acute kidney injury, especially in older adults or those with pre-existing kidney disease. Symptoms: reduced urine output, swelling in legs or ankles, fatigue, shortness of breath. Stay hydrated aggressively during dose escalation; call your doctor if you cannot keep fluids down for 24+ hours.
Diabetic Retinopathy Progression
In patients with pre-existing diabetic retinopathy, rapid glucose improvement from any GLP-1 treatment can paradoxically worsen retinopathy short-term. Symptoms: blurred vision, floaters, dark spots, or vision loss. A dilated eye exam before starting and at regular intervals is standard of care for diabetics on tirzepatide.
NAION (Sudden Vision Loss), 2025 Label Consideration
In 2024-2025, research linked GLP-1 drugs to a higher risk of non-arteritic anterior ischemic optic neuropathy (NAION), a sudden and usually painless vision loss in one eye caused by blocked blood flow to the optic nerve. The FDA added NAION to Ozempic's label in 2025, and tirzepatide is under the same class scrutiny. Any sudden vision change, especially in one eye, needs same-day ophthalmology evaluation.
Ileus (Intestinal Paralysis)
The FDA added ileus to the Ozempic and tirzepatide label areas in 2023 after post-marketing reports of severe intestinal dysmotility. Symptoms: no bowel movements for several days, severe abdominal distension, persistent vomiting. Ileus is a medical emergency, stop the drug and go to the ER.
Tirzepatide Lawsuits: The Gastroparesis and NAION MDL
Important context for anyone researching the long-term safety picture.
Tirzepatide (Mounjaro/Zepbound) is named alongside Ozempic in a multidistrict litigation (MDL) consolidating thousands of lawsuits against Novo Nordisk and Eli Lilly. The core allegations: inadequate warning about severe gastroparesis and related GI complications from GLP-1 and GLP-1/GIP drugs. Separate complaints address NAION-linked vision loss.
Regulatory timeline:
- September 2023: FDA added ileus warning to GLP-1 labeling
- 2024: FDA reviewed suicidal ideation reports for GLP-1 drugs; review did not establish causation
- 2025: FDA added NAION information to Ozempic (semaglutide) prescribing information; tirzepatide labeling under ongoing class review
- 2026: MDL cases ongoing in federal court; outcomes pending
None of this means tirzepatide is definitively unsafe. It does mean the post-marketing risk profile at scale, particularly severe GI and rare ocular events, is more fully understood in 2026 than it was at approval.
Tirzepatide vs Semaglutide vs Retatrutide: Side Effect Comparison
If you're trying to decide between these three, or if you've already been on semaglutide and are switching, this table matters. The mechanisms are different enough that the side effect profiles diverge in meaningful ways.
| Side Effect | Tirzepatide (GIP+GLP-1) | Semaglutide (GLP-1 only) | Retatrutide (GIP+GLP-1+Glucagon) |
|---|---|---|---|
| Nausea | 25-45% | 20-44% | 40-57% |
| Vomiting | 9-25% | 5-24% | 18-36% |
| Diarrhea | 20-30% | 15-30% | ~25-35% |
| Constipation | 16-29% | 5-24% | ~20-25% |
| Decreased appetite | Strong | Moderate-strong | Very strong |
| HR increase | +2-4 bpm | +1-2 bpm | +2-5 bpm |
| Injection site reactions | 3-7% | 3-6% | Similar |
| Thyroid warning | Black box | Black box | Likely (same class) |
| Weight loss (max dose trial) | ~22% body weight | ~15-17% body weight | ~24% body weight |
| Availability (research) | Limited (compounded) | Available (S-5 via Ascension) | Available (R-30 via Ascension) |
Practically: tirzepatide and semaglutide have comparable GI side effect rates, but tirzepatide produces better weight loss. Retatrutide, the triple receptor agonist, has higher nausea rates than both but even more aggressive fat loss in Phase 2 trials. For a deeper look at retatrutide's specific side effect profile, see our retatrutide side effects guide.
Tirzepatide Side Effects Timeline: When They Start, Peak, and Stop
One of the most common questions people have before starting: when does the nausea actually stop? Here's the realistic timeline based on trial data and clinical experience.
| Phase | Timing | What's Happening | Typical Duration |
|---|---|---|---|
| Initial adjustment | Weeks 1-4 (2.5mg) | Light nausea, possible loose stools. Body adjusting to gastric slowing. | Fades by week 3-4 for most |
| First escalation | Weeks 5-8 (5mg) | Nausea returns with dose increase. Peak GI discomfort for most people. | 2-4 weeks then subsides |
| Mid escalation | Weeks 9-16 (7.5-10mg) | GI tolerance significantly improved. Constipation may replace nausea. | Nausea rare; constipation ongoing |
| Higher doses | Weeks 17-24 (12.5-15mg) | Some nausea may return with escalation but usually milder than early phases. | 1-2 weeks per step |
| Hair shedding | Months 2-5 | Telogen effluvium from rapid weight loss. Not a direct drug effect. | Resolves as weight stabilizes |
| Stable dosing | Month 6+ | Most GI side effects minimal or absent. Body fully adapted. | Ongoing at low background level |
The pattern most people experience: rough first 4-6 weeks, another rough patch with each dose step, then sustained tolerance once a maintenance dose is reached. If side effects are still severe at 12+ weeks on the same dose without recent escalation, that's worth flagging with your prescriber, it falls outside the typical pattern.
How to Minimize Tirzepatide Side Effects
Most people who successfully stay on tirzepatide long-term say the same things: they took it slow, they changed how they ate, and they didn't push through severe symptoms. Here's what the evidence and anecdotal experience both support:
Dietary Adjustments
- Eat smaller meals, 4-6x per day instead of 3 large ones
- Avoid high-fat, greasy, or fried foods, they worsen nausea dramatically
- Eat slowly. 20+ minutes per meal. This sounds annoying; it actually works
- Cut alcohol, especially during the first 4-8 weeks
- Add high-fiber foods gradually to reduce constipation/diarrhea swings
- Stay hydrated, especially if experiencing diarrhea or vomiting
Timing and Administration
- Inject on the same day each week, consistency reduces hormonal variability
- Some people find injecting in the evening reduces daytime nausea
- Don't inject into scar tissue or areas with lipohypertrophy
- Let the pen reach room temperature before injecting
Dose Escalation Strategy
The standard escalation schedule is 4 weeks at each dose. There's no rule that says you must escalate at 4 weeks. If you're still struggling with GI symptoms at week 6 or 8, staying at 5mg for another month before going to 10mg is completely reasonable, and likely results in better long-term adherence.
Medications That Help
- Ondansetron (Zofran): Prescription antiemetic, highly effective for tirzepatide-induced nausea
- Metoclopramide: Helps gastric motility, reduces nausea and bloating
- Famotidine (Pepcid): OTC, helps GERD symptoms
- Psyllium husk (Metamucil): Helps both diarrhea and constipation by normalizing stool consistency
- Ginger supplements or tea: Some evidence for reducing nausea severity
Mounjaro vs Zepbound Side Effects: Same Drug, Different Label
Mounjaro and Zepbound are both tirzepatide. The side effect profile is identical in kind, but the approved use and dose range differ.
- Mounjaro: FDA-approved for type 2 diabetes. Same dose range as Zepbound.
- Zepbound: FDA-approved for chronic weight management in adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related condition.
- Dose range: 2.5mg, 5mg, 7.5mg, 10mg, 12.5mg, 15mg weekly for both, titrated over 4-6 months
- Side effect profile: Identical. Any GI, serious, or long-term effect documented for one applies to the other.
- Insurance coverage: Mounjaro is widely covered for diabetes; Zepbound coverage for weight loss varies heavily by plan and often requires BMI documentation.
If you tolerate one, you will tolerate the other. If Mounjaro caused you problems, switching to Zepbound will not change that, only the prescribing indication and insurance picture changes.
Side Effects Specific to Women
- Menstrual changes: Irregular cycles or changes in flow are common during significant weight loss
- Hair loss: Reported more frequently by women, likely a combination of reporting bias and hair sensitivity to rapid caloric change
- Fertility shifts: Weight loss can restore ovulation in PCOS patients, sometimes leading to unexpected pregnancies. Reliable contraception is essential because tirzepatide is not recommended in pregnancy.
- Oral contraceptive absorption: Slowed gastric emptying from tirzepatide can reduce oral contraceptive effectiveness; the FDA recommends using a non-oral contraceptive method or adding a barrier method for 4 weeks after starting and after each dose increase.
- Breastfeeding: Not recommended; tirzepatide transfer into breast milk has not been fully characterized
Tirzepatide Pregnancy and Breastfeeding Warnings
Tirzepatide is not recommended during pregnancy.
Early toxicology data show adverse developmental outcomes at clinically relevant exposures. Human pregnancy data is limited. The FDA-approved labeling recommends discontinuing tirzepatide at least 2 months before a planned pregnancy to allow the drug to clear the body (tirzepatide half-life is ~5 days; full clearance takes about 4-5 weeks).
If pregnancy is confirmed while on tirzepatide, most clinicians will stop it immediately and discuss glucose management or weight-related alternatives with an OB/GYN or endocrinologist.
Tirzepatide Drug Interactions
Tirzepatide doesn't have a large number of dangerous drug interactions, but the ones that exist are clinically important, particularly for people managing multiple conditions alongside weight loss or diabetes.
| Drug / Drug Class | Interaction | What to Do |
|---|---|---|
| Insulin | Increased hypoglycemia risk when combined. SURPASS-5 trial: ~19% hypoglycemia rate. | Insulin dose often needs reduction when starting tirzepatide. Work with prescriber. |
| Sulfonylureas (glipizide, glyburide) | Combined GI effect amplifies hypoglycemia risk | Sulfonylurea dose reduction typically required |
| Oral medications (general) | Gastric slowing delays absorption of all oral drugs taken around the same time | Take time-sensitive medications (thyroid, anticoagulants) at consistent times, ideally not immediately post-injection |
| Warfarin / anticoagulants | Delayed absorption may affect INR stability, especially early in treatment | Monitor INR more closely during initiation and dose escalation |
| Oral contraceptives | Gastric slowing may reduce absorption reliability of estrogen/progestin pills | Consider backup contraception during initiation and escalation phases |
| Other GLP-1 agonists | Additive GI effects; no established benefit to combining; increases adverse event risk | Do not combine tirzepatide with semaglutide, liraglutide, or other GLP-1 drugs |
Always provide your complete medication list to your prescriber before starting tirzepatide. The gastric emptying effect on oral drug absorption is a class-wide issue with GLP-1 agonists that can affect medications you might not expect, particularly anything where timing and peak concentration matters.
When to Stop Tirzepatide: Side-Effect Red Flags
There's a difference between side effects that are uncomfortable but manageable, and side effects that are genuinely dangerous. Here's the line:
- Severe persistent abdominal pain, especially radiating to the back (possible pancreatitis)
- Signs of allergic reaction: throat swelling, difficulty breathing, rash with hives
- Inability to keep any fluids down for more than 24 hours
- Symptoms of hypoglycemia that don't resolve with glucose intake
- New neck lump, hoarseness, or difficulty swallowing
- Severe vision changes (rare, but reported in diabetic patients)
If you're stopping tirzepatide due to intolerable but non-emergency side effects, there's no need to taper, you can simply stop. Unlike some medications, tirzepatide doesn't cause physiological withdrawal. Weight regain can happen relatively quickly after stopping, though.
[Image #1]
What Happens When You Stop Tirzepatide
The realistic picture for people considering stopping.
- Drug clearance: Half-life ~5 days; full clearance from the body takes 4-5 weeks
- Appetite return: Food noise and hunger return within 2-4 weeks as the drug clears
- Weight regain: SURMOUNT-4 extension data showed roughly 14% of the lost weight returned within 1 year of stopping in people who did not have a structured maintenance plan; the relative regain can reach two-thirds of lost weight in people without lifestyle anchors in place
- GI symptoms resolve: Stomach emptying rates return to normal; food tolerance improves
- Glucose control: In diabetics, A1c typically returns toward pre-treatment levels if no other diabetes treatment is active
- Maintenance options: Lower-dose tirzepatide (sometimes called "micro-dose maintenance"), switching to a weaker GLP-1, or structured nutrition plus resistance training are the main paths
References and FDA Resources
- FDA prescribing information for Mounjaro and Zepbound (tirzepatide): accessdata.fda.gov
- FDA MedWatch adverse event reporting: 1-800-FDA-1088 or FDA.gov/MedWatch
- SURPASS-1 through SURPASS-5 trial publications (tirzepatide diabetes program)
- SURMOUNT-1 through SURMOUNT-4 trial publications (tirzepatide weight management program)
- FDA ileus label addition: September 2023
- FDA NAION-related updates: 2025 class review ongoing for tirzepatide; NAION added to semaglutide labeling in 2025